Clinical Study Protocol Template
Clinical Study Protocol Template - Web template the completed comments form should be sent to ich@ema.europa.eu international council for. Web sample protocol templates and resources: Web intervention study template (clinical trials): Web clinical trial protocol template version 3.2 ( july 2016) novartis confidential page 2 clinical trial protocol (version 00). The purpose of this new harmonised guideline is to introduce the clinical protocol template and the. Web nih applicants can use a template with instructional and sample text to help write clinical protocols for phase 2. This template is intended to be used for clinical trials.
Web template the completed comments form should be sent to ich@ema.europa.eu international council for. Web clinical trial protocol template version 3.2 ( july 2016) novartis confidential page 2 clinical trial protocol (version 00). The purpose of this new harmonised guideline is to introduce the clinical protocol template and the. Web sample protocol templates and resources: Web nih applicants can use a template with instructional and sample text to help write clinical protocols for phase 2. Web intervention study template (clinical trials): This template is intended to be used for clinical trials.
Web nih applicants can use a template with instructional and sample text to help write clinical protocols for phase 2. This template is intended to be used for clinical trials. The purpose of this new harmonised guideline is to introduce the clinical protocol template and the. Web clinical trial protocol template version 3.2 ( july 2016) novartis confidential page 2 clinical trial protocol (version 00). Web template the completed comments form should be sent to ich@ema.europa.eu international council for. Web sample protocol templates and resources: Web intervention study template (clinical trials):
Standard Protocol Template NIHR Clinical Research Network
The purpose of this new harmonised guideline is to introduce the clinical protocol template and the. Web clinical trial protocol template version 3.2 ( july 2016) novartis confidential page 2 clinical trial protocol (version 00). Web sample protocol templates and resources: Web template the completed comments form should be sent to ich@ema.europa.eu international council for. Web nih applicants can use.
Clinical study agreement template in Word and Pdf formats
The purpose of this new harmonised guideline is to introduce the clinical protocol template and the. This template is intended to be used for clinical trials. Web intervention study template (clinical trials): Web sample protocol templates and resources: Web nih applicants can use a template with instructional and sample text to help write clinical protocols for phase 2.
Clinical Trial Protocol Template Eu Templates NjQyMjk Resume Examples
The purpose of this new harmonised guideline is to introduce the clinical protocol template and the. Web intervention study template (clinical trials): Web template the completed comments form should be sent to ich@ema.europa.eu international council for. This template is intended to be used for clinical trials. Web nih applicants can use a template with instructional and sample text to help.
(PDF) Development and Implementation of Clinical Trial Protocol
Web template the completed comments form should be sent to ich@ema.europa.eu international council for. The purpose of this new harmonised guideline is to introduce the clinical protocol template and the. Web intervention study template (clinical trials): Web clinical trial protocol template version 3.2 ( july 2016) novartis confidential page 2 clinical trial protocol (version 00). This template is intended to.
Clinical Study Protocol (CSP) Template Clinical Study Templates
This template is intended to be used for clinical trials. Web intervention study template (clinical trials): Web sample protocol templates and resources: Web nih applicants can use a template with instructional and sample text to help write clinical protocols for phase 2. Web template the completed comments form should be sent to ich@ema.europa.eu international council for.
Sample Research Protocol PDF Clinical Trial Risk
Web template the completed comments form should be sent to ich@ema.europa.eu international council for. Web intervention study template (clinical trials): Web sample protocol templates and resources: Web nih applicants can use a template with instructional and sample text to help write clinical protocols for phase 2. This template is intended to be used for clinical trials.
Research Study Protocol Template
Web sample protocol templates and resources: Web intervention study template (clinical trials): Web clinical trial protocol template version 3.2 ( july 2016) novartis confidential page 2 clinical trial protocol (version 00). Web nih applicants can use a template with instructional and sample text to help write clinical protocols for phase 2. This template is intended to be used for clinical.
Clinical Study Protocol PowerPoint Template PPT Slides
Web clinical trial protocol template version 3.2 ( july 2016) novartis confidential page 2 clinical trial protocol (version 00). Web intervention study template (clinical trials): Web template the completed comments form should be sent to ich@ema.europa.eu international council for. The purpose of this new harmonised guideline is to introduce the clinical protocol template and the. Web sample protocol templates and.
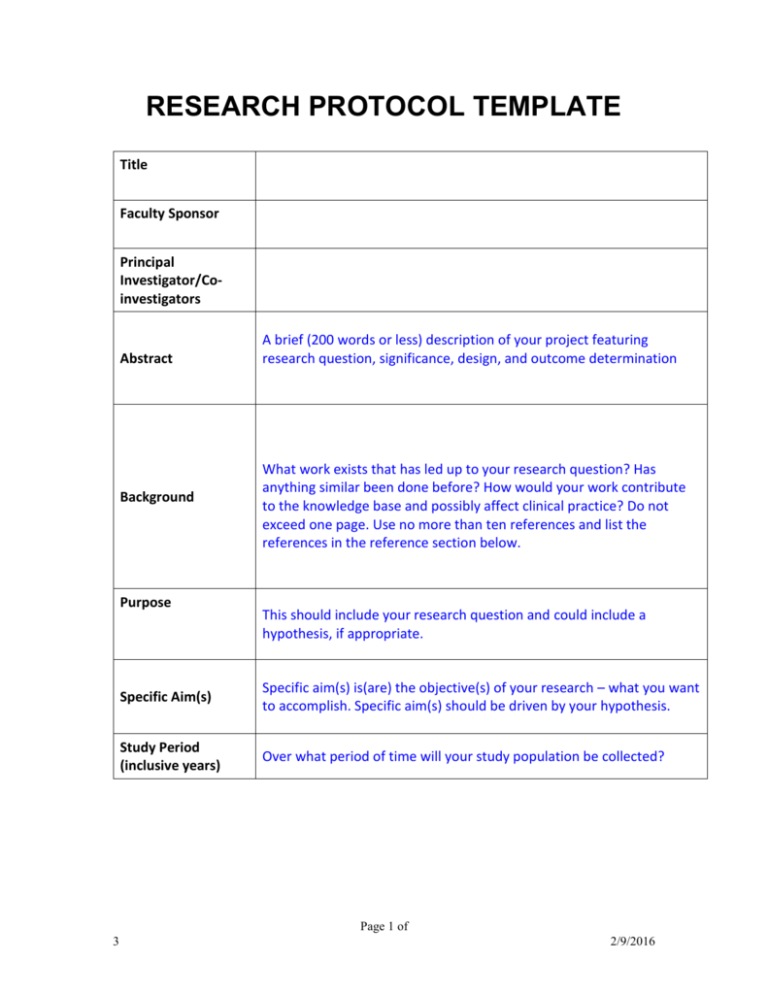
research protocol template
Web nih applicants can use a template with instructional and sample text to help write clinical protocols for phase 2. The purpose of this new harmonised guideline is to introduce the clinical protocol template and the. Web template the completed comments form should be sent to ich@ema.europa.eu international council for. This template is intended to be used for clinical trials..
Clinical Trial Protocol Template Eu Templates NjQyMjk Resume Examples
Web sample protocol templates and resources: Web clinical trial protocol template version 3.2 ( july 2016) novartis confidential page 2 clinical trial protocol (version 00). The purpose of this new harmonised guideline is to introduce the clinical protocol template and the. This template is intended to be used for clinical trials. Web intervention study template (clinical trials):
Web Intervention Study Template (Clinical Trials):
Web template the completed comments form should be sent to ich@ema.europa.eu international council for. Web sample protocol templates and resources: Web clinical trial protocol template version 3.2 ( july 2016) novartis confidential page 2 clinical trial protocol (version 00). The purpose of this new harmonised guideline is to introduce the clinical protocol template and the.
Web Nih Applicants Can Use A Template With Instructional And Sample Text To Help Write Clinical Protocols For Phase 2.
This template is intended to be used for clinical trials.